Current Report Filing (8-k)
26 6월 2014 - 8:03PM
Edgar (US Regulatory)
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(D)
of the Securities Exchange Act of 1934
June 26, 2014
Date of report (Date of earliest event reported)
Agile Therapeutics, Inc.
(Exact name of registrant as specified in its charter)
|
Delaware |
|
001-36464 |
|
23-2936302 |
|
(State or other jurisdiction
of incorporation) |
|
(Commission
File Number) |
|
(IRS Employer
Identification No.) |
|
101 Poor Farm Road
Princeton, New Jersey
(Address of principal executive offices) |
|
08540
(Zip Code) |
Registrant’s telephone number, including area code (609) 683-1880
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
o Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425).
o Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12).
o Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)).
o Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c))
Item 7.01 Regulation FD Disclosure
On June 26, 2014, Al Altomari, Chief Executive Officer and President of Agile Therapeutics, Inc. (the “Company”), and Scott Coiante, the Company’s Vice President and Chief Financial Officer, participated at the Janney Capital Markets Boston Healthcare 1X1 Corporate Access Day in Boston, MA. The Company is furnishing a copy of the presentation used at this conference, which is attached as Exhibit 99.1.
In accordance with General Instructions B.2 and B.6 of Form 8-K, the information included in this Current Report on Form 8-K (including Exhibit 99.1 hereto), shall not be deemed “filed” for the purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference into any filing made by the Company under the Exchange Act or Securities Act of 1933, as amended, except as shall be expressly set forth by specific reference in such a filing.
Item 9.01. Financial Statements and Exhibits.
(d) Exhibits.
|
Exhibit
Number |
|
Description |
|
99.1 |
|
Agile Therapeutics, Inc. Presentation |
2
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
|
|
Agile Therapeutics, Inc. |
|
|
|
|
|
|
|
Dated: June 26, 2014 |
By: |
/s/ Alfred Altomari |
|
|
Name: |
Alfred Altomari |
|
|
Title: |
President and Chief Executive Officer |
3
Exhibit 99.1
|

|
Agile
Therapeutics Rule.pngNot for promotional purposes
|
|

|
2
Rule.pngProperty of Agile Therapeutics –06.26.2014 Forward-Looking Statement
Certain information contained in this presentation and other matters
discussed today or answers that may be given in response to questions may
include “forward-looking statements”.We may, in some cases, use terms such as
“predicts,” “believes,” “potential,” “continue,” “estimates,” “anticipates,”
“expects,” “plans,” “intends,” “may,” “could,” “might,” “will,” “should” or
other words that convey uncertainty of future events or outcomes to identify
these forward-looking statements.In particular, the Company’s statements
regarding trends and potential future results are examples of such
forward-looking statements. The forward-looking statements are subject to
important factors, risks and uncertainties, including, but not limited to,
the success, timing and cost of our ongoing clinical trials and anticipated
clinical trials for our current product candidates, including statements
regarding the timing of initiation and completion of the trials; the timing
of and our ability to obtain and maintain U.S. Food and Drug Administration
or other regulatory authority approval of, or other action with respect to,
our product candidates; the Company’s ability to obtain the capital necessary
to fund its operations; the Company’s ability to generate revenues; the
successful implementation of the Company’s research and development programs
and collaborations; the acceptance by the market of the Company’s products;
the Company’s ability to successfully defend its intellectual property or
obtain the necessary licenses at a cost acceptable to the Company, if at all;
the success of the Company's license agreements; and other factors, including
general economic conditions and regulatory developments, not within the
Company’s control.These factors could cause actual results and developments
to be materially different from those expressed in or implied by such
statements.The forward-looking statements are made only as of the date of
this presentation and the Company undertakes no obligation to publicly update
such forward-looking statements to reflect subsequent events or circumstance.
|
|

|
3 Rule.png Property
of Agile Therapeutics –06.26.2014 Agile Investment Thesis •$4.2 Billion
combined hormonal contraceptive (CHC) market •Significant Unmet Need: No
low-dose CHC patch on the market today Large Market Opportunity •Over 1,500
women have received Twirla in clinical trials that showed favorable safety
and tolerability •Two completed randomized phase 3 trials showed pregnancy
rate comparable to comparator pills Significant Clinical Experience •CRL and
FDA communications provide clear guidance on path forward with one single-arm
trial •Potential for near-term approval in late 2016 Clear Regulatory Path
•Wholly owned assets means company is free to partner or sell •Company can
market directly through focused sales force Multiple Strategic Options •Deep
experience in women’s health and contraceptive products World Class Team •6
issued patents, protection to 2028 •Technological and manufacturing know-how
High Barriers to Generic Entry
|
|

|
4
Rule.pngProperty of Agile Therapeutics –06.26.2014 Agile Executive Management
Team Al Altomari President and Chief Executive Officer Scott Coiante Vice
President and Chief Financial Officer Elizabeth Garner, M.D.,M.P.H. Sr. Vice
President and Chief Medical Officer Katie MacFarlane, Pharm. D. Chief
Commercial Officer Deep Experience in Women’s Healthcare and Contraceptive
Products
|
|

|
5
Rule.pngProperty of Agile Therapeutics –06.26.2014 Source: IMS NSP, retail +
non-retail, Dec 2013 Loestrin is a registered trademark of Actavis. Evra and
Micronor are registered trademarks of Johnson & Johnson Depo-Provera is a
registered trademark of Pfizer Agile products are designed to provide
convenience and facilitate compliance Contraceptive Market is a Large
Opportunity Twirla™ AG200AG200--ERER AG200AG200--SPSP Ring
ortho_evra_patch.jpgPatches Pills C:UsersKatiePicturesLoestrin
photo.jpgCombination Hormonal Contraceptive Market $4.2 Billion AG890AG890
Implant C:UsersKatiePicturesImplanon photo.pngDepo Provera.jpgInjection
C:UsersKatiePicturesMirena photo.pngIUD Pills Progestin-only
Contraceptive Market $1.4 Billion
http://www.bd.nl/polopoly_fs/1.4167231.1390569730!/image/image.JPG_gen/derivatives/landscape_800_600/image-4167231.JPG
|
|

|
6
Rule.pngProperty of Agile Therapeutics –06.26.2014 CHC = Combined Hormonal
Contraception, P-Only = Progestin-Only Contraception = Additional patch
development to determine optimal formulation and dose = Indicates phase to
enter following patch development Agile Has Four Products in Development CHC
Market TwirlaTM AG200-ER AG200-SP AG890 P-Only Market Development Phase I
Phase II Phase III
|
|

|
7
Rule.pngProperty of Agile Therapeutics –06.26.2014 •Contains the active
ingredients levonorgestrel (LNG) and ethinyl estradiol (EE), which have over
25 years of history of use in contraceptives –LNG is used as a standard for
comparison of VTE risk among progestins –EE is the syntheticestrogen in most
currently marketed contraceptives •Agile’s proprietary Skinfusion®technology
provides hormone delivery in an appealing form Twirla is a Low-Dose
Contraceptive Patch C:UsersKatieDropboxAgileCorporate Info, LogosLogos
and ImagesTwirlaTWIRLA_Logo-TM_G.jpgPeripheral adhesive system is designed
to allow patch adherence and patient comfort and is intended to prevent the
active and inactive ingredients from migrating to the outer edges Inner,
active matrix adhesive system delivers both EE and LNG at targeted levels
through the skin
|
|

|
8
Rule.pngProperty of Agile Therapeutics –06.26.2014 Skinfusion Technology in
Twirla C:UsersKatieDropboxAgileCorporate Info, LogosLogos and
ImagesTwirlaTWIRLA_Logo-TM_G.jpgTwirla is <1mm in thickness Peripheral
Backing(soft, silky fabric) PeripheralAdhesiveSystem Active Matrix Integrated
into a single patch Release Liner
|
|

|
9
Rule.pngProperty of Agile Therapeutics –06.26.2014 Agile
Exclusivity/Competition Strategies for Twirla Expected Hatch-Waxman
exclusivity (3 years) 6 issued patents to list in Orange Book •Issued patents
expire in 2021 (4) & 2028 (2) •Additional patents under prosecution
Limited patch manufacturers •Specialized equipment Transdermal know-how
Agilepipeline / line extensions C:UsersKatiePicturesOrange Book
logo.pngC:UsersGregory ArnoldDesktopNew folderBldg 50 Sign 31October2008.JPGC:UsersKatieDropboxAgilePresentationsCorporate
PresentationCorium and ManufacturingPictures
22April2014IMG_0822[1].JPGhttp://www.azbio.org/pinniped/wp-content/uploads/2012/02/fdalogodhhs.jpg
|
|

|
10
Rule.pngProperty of Agile Therapeutics –06.26.2014 SUN MON TUE WED THU FRI
SAT 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27
28 29 30 31 How Would Women use Twirla? •21/7-day regimen like many birth
control pills –Women apply a patch once-a-week for three weeks followed by
4th week without a patch •Can be applied to abdomen, buttock, or upper torso
C:UsersKatieDropboxAgileCorporate Info, LogosLogos and
ImagesTwirlaTWIRLA_Logo-TM_G.jpgButtock 48% Abdomen 40% Source: Data on
File, Agile Therapeutics Upper Torso 12% 7days no patch Patch 1 Patch 2 Patch
3 Patch 1
|
|

|
11
Rule.pngProperty of Agile Therapeutics –06.26.2014 Ortho Evra quickly gained
share: the most successful contraceptive launch Ortho Evra –A Meteoric Rise
•Most successful contraceptive launch in history •Reached TRx share of 11%
and nearly $400 million in annual sales in 2004 •Labeled as 20mg/day EE
•Sales hampered by supply constraints and a precipitous fall •Thromboembolic
events (VTEs) reported to FDA •Study published in 2005 showing higher EE levels
than ring and low-dose pill •Bolded warning added to Evra label in Nov 2005
•Johnson & Johnson stopped active promotion •Mylan launched Evra generic
in April 2014 0.0% 2.0% 4.0% 6.0% 8.0% 10.0% 12.0% Apr-02 Jul-02 Oct-02
Jan-03 Apr-03 Jul-03 Oct-03 Jan-04 Apr-04 TRx Share (%) 0.0% 2.0% 4.0% 6.0%
8.0% 10.0% 12.0% Sep-04 Mar-05 Sep-05 Mar-06 Sep-06 Mar-07 Sep-07 Mar-08
Sep-08 Mar-09 TRx Share (%) Evra Contraceptive Patch History supply
constraints label change Sources: IMS NPA and NSP Ortho Evra Package Insert
and van den Heuvel, Contraception 2005;72:168-174 Validated MarketMarket
|
|

|
12
Rule.pngProperty of Agile Therapeutics –06.26.2014 Characteristic Form of
product Transdermal patch Round, approximately 28 square centimeters Soft,
silky, stretchy fabric Transdermal patch Square, approximately 20 square
centimeters Smooth, plastic film Active ingredients EE, LNG EE,
norelgestromin Pharmacokinetic profile EE delivered per day ~30 micrograms
60% higher than that of an oral contraceptive containing 35 micrograms (~56
micrograms)** Regimen One patch weekly 21 days active/7 days patch-free Same
as Twirla Package configurations 1 box of 3 patches = 1 cycle 1 box with 1
patch = replacement Same as Twirla Top four adverse events/reactions in
clinical trials Nausea 3.0% Application site irritation 2.4% Breast
tenderness 2.1% Headache 2.0% Breast symptoms 22.4% Headache 21.0%
Application site disorders 17.1% Nausea 16.6% Twirla Product Profile Compared
to Ortho Evra *Information is based upon the characteristics of Twirla and
other Twirla attributes observed in our Phase 3 clinical trials andthe
currently marketed Evra product label and publicly-available information. We
have not performed a head-to-head comparison of Twirla to Evra. **The Evra
package insert indicates a strength of 35µg EE per day
C:UsersKatieDropboxAgileCorporate Info, LogosLogos and
ImagesTwirlaTWIRLA_Logo-TM_G.jpgHand-092_b.jpgComparison of Twirla and Evra
Product Characteristics* Screen shot 2012-07-10 at 3.25.38 PM.pngOrtho Evra
LogoEvra is a registered trademark of Johnson & Johnson
|
|

|
13
Rule.pngProperty of Agile Therapeutics –06.26.2014 EE Concentrations (pg/ml)
EE (pg/mL) Patch Change Patch Removal 140 120 100 80 60 40 20 0 15 25 16 17
18 19 20 21 22 23 24 Day (Week three of Cycle) OC 35µg –Days 15-20
(estimated) Twirla Patch –Days 15-21 (observed) OC 35µg –Day 21 (observed)
Evra –Days 15-21 (from label) Sources: Archer, et al. Contraception
2012;85:595-601 Archer, at al. Oral presentation at American Society of
Reproductive Medicine (ASRM), 2010 Twirla PK Profile Compared to 35µg Oral
Contraceptive and Ortho Evra Lower dose of EE from Twirla compared to Evra
Data are not a head-to-head comparison of Twirla to Evra. The Evra curve was
estimated based on the graph provided in the Evra label.
|
|

|
14
Rule.pngProperty of Agile Therapeutics –06.26.2014 The Path Forward –New
TwirlaTrial •CRO lacked experience with large contraceptive trials •Poor
study conduct at several clinical sites •Study population at higher risk for
non-compliance •No utilization of technology •New Team / Chief Medical
Officer -Dr. Elizabeth Garner •New top-tier CRO -Parexel •Data-driven site
selection •Rigorous screening of subjects •Use of technology for improved
study oversight –PHT e-diary What we believe were the issues Why a new trial
can be successful
|
|

|
15
Rule.pngProperty of Agile Therapeutics –06.26.2014 Twirla Product Development
Summary Comprehensive clinical program enrolled over 2,100 women –Over 1,500
women have received Twirla –485 women have received Twirla for 12 months
Pharmacokinetic profile is consistent with a low-dose contraceptive*
Effectiveness in Phase 3 studies generally comparable to approved low- dose
oral contraceptive (OC) comparators** Twirla was well-tolerated with low rate
of estrogen-associated adverse events** The Skinfusion technology performed
well with daily activities and conditions, including showering, exercise,
swimming, and heat/humidity *Archer DF, et al, Contraception 2012
Jun;85(6):595-601 **Kaunitz A, et al, Obstetrics and Gynecology 2014
Feb;123(2):1-10
|
|

|
16
Rule.pngProperty of Agile Therapeutics –06.26.2014 Twirla Phase 3 Clinical
ResultsPrimary Effectiveness Measure •Pearl Index (PI) = Pregnancies per 100
women-years of product use *Pooled data from 2 clinical trials, CL-12 and
CL-13. Data on File, Agile Therapeutics, CRL response to FDA, Aug 2013.
**Quartette package insert. Quartette is a registered trademark of Teva, Inc.
Observed PIs in Phase 3 Trials Twirla Phase 3 Trials* Pill 6.72 Twirla 5.76
HighestPI approved to date** 3.19 Number of On-drug pregnancies Number of
28-day On-drug cycles (13) (100)
|
|

|
17
Rule.pngProperty of Agile Therapeutics –06.26.2014 Impact of New Users and
Minorities on Twirla PI •We believe clinical results were affected by study
conduct issues at several sites –36% of on-drug pregnancies reported at 4 of
96 sites* •Study population comprised high proportion of new users and
minorities who are known to be at higher risk of non-compliance and
pregnancy** –New users had ~3 times higher non-compliance than experienced
users –These factors impacted the observed PIs Twirla PI Stratified by New
Users and Minority Subjects Current usersa 0.0 Experienced usersb 3.0 New
usersc 8.7 Black subjects who were new users 16.0 (a) Current users = subjects
who used a hormonal contraceptive within seven days of enrollment (b)
Experienced users = recent (used a hormonal contraceptive within 6 months of
enrollment) and current users (c) New users = never used hormonal
contraception (HC) or had not used HC in the 6 months prior to enrollment
*These 4 sites represented 15% of the randomized subject population
**Hatcher, et al. Contraceptive Technology 20thEd, 2011, page 50 and Pooled
data from 2 clinical trials, CL-12 and CL-13. Data on File, Agile Therapeutics,
CRL response to FDA, Aug 2013
|
|

|
18
Rule.pngProperty of Agile Therapeutics –06.26.2014 Twirla Regulatory
Interactions and Path Forward *Pooled data from 2 clinical trials, CL-12 and
CL-13. Data on File, Agile Therapeutics, CRL response to FDA, Aug 2013 2014
2013 2012 2011 2010 Two Phase 3 Trials Conducted •Effectiveness comparable to
low dose OC comparators* NDA Filed CRL Received FDA Requested New Phase 3
Study •PI results not sufficient for approval •Study conduct issues FDA
Interactions •Agile agreed to new study with simplified design and improved
conduct •FDA provided clear guidance on requirements for new Phase 3 trial
Initiate New Phase 3 Trial
|
|

|
19
Rule.pngProperty of Agile Therapeutics –06.26.2014 Planned Twirla Phase 3
Trial •Single-arm, open-label study •~2,000 sexually active subjects will
receive Twirla for up to one year •50 to 70 sites U.S. sites NewNew Twirla
TrialTwirla Trial CRO - Parexel The best possible partner Site Selection
Data-driven site qualification Compliant Subjects Rigorous screening
Execution Achieve high retention and low loss to follow-up •Robust technology
•Real-time data •Experienced study coordinators •Experienced users
•Representative demographics •Extensive training •Electronic diaries and
reminders Pregnancy Adjudication Accurate and Timely •Pregnancy Review
Committee
|
|

|
20
Rule.pngProperty of Agile Therapeutics –06.26.2014 Twirla Value Proposition
•Patch designed to offer convenience and compliance –Physicians want products
that offer their patients convenience .give them confidence in compliance
•Selection of hormones at right dose –Levonorgestrel is one of the progestins
with the lowest risk of VTE –Over 25 years of market experience –Delivers low
daily dose (~ 30µg) of ethinyl estradiol •Has unique selling proposition for
patients –Offers convenience and compliance –Fits with the busy lifestyle of
today’s women Right product and right dose for physicians and patients
C:UsersKatieDropboxAgileCorporate Info, LogosLogos and
ImagesTwirlaTWIRLA_Logo-TM_G.jpgSources: Contraceptive Patch Assessment
Studies (n=152 ObGyns and n=307 consumers), Kantar Health, Dec 2010 and
Twirla ObGyn/NP and Consumer Market Research, RG&A, Aug 2012
|
|

|
21
Rule.pngProperty of Agile Therapeutics –06.26.2014 Agile Has Additional
Products in Development Product Rationale Development •Standard contraceptive
regimen •Validated market opportunity •Currently in Phase III •Responding to
CRL AG200-ER Extended Cycle Regimen •Fewer periods per year •Potential
advantage over OC regimens •No new product development required •Potential to
progress into Phase 3 in 2015 AG200-SP Shortened Hormone- Free Interval
(SHFI) •Shorter, lighter periods •Potential to improve contraceptive
effectiveness •Requires product development •Potential to initiate Phase I in
2015 AG890 Progestin-OnlyRegimen •Designed for women who are unable or
unwilling to take estrogen •Initial Phase I/II trial conducted •Additional
product development required C:UsersKatieDropboxAgileCorporate Info,
LogosLogos and ImagesTwirlaTWIRLA_Logo-TM_G.jpg
|
|

|
22
Rule.pngProperty of Agile Therapeutics – 06.26.2014 Source: IMS sales retail
+ non-retail, Dec 2013 Loestrin and Minastrin a registered trademarks of
Actavis (formerly Warner-Chilcott), Ortho Evra and Tri-Cyclen are registered
trademarks of J&J, Yaz is a registered trademark of Bayer, Nuvaring is a
registered trademark of Merck Combination Hormonal Contraceptive Market is a
Large Opportunity Leading Brands Form 2013 Sales ($Millions) Nuvaring (Merck)
Ring $569 Tri-Cyclen-Lo (Johnson & Johnson) Pill $469 Loestrin/Minastrin
24 (Actavis/Warner Chilcott) Pill $413 LoLoestrin (Actavis/Warner Chilcott)
Pill $272 Evra (Johnson & Johnson) Patch $152 Beyaz (Bayer) Pill $111
Generess (Actavis) Pill $82 Yaz (Bayer) Pill $53 TwirlaTwirla™™
AG200AG200--ERER AG200AG200--SPSP Ring ortho_evra_patch.jpgPatches Pills
C:UsersKatiePicturesLoestrin photo.jpgCombination Hormonal Contraceptive
Market $4.2 Billion
http://www.bd.nl/polopoly_fs/1.4167231.1390569730!/image/image.JPG_gen/derivatives/landscape_800_600/image-4167231.JPG
|
|

|
23
Rule.pngProperty of Agile Therapeutics – 06.26.2014 CHC Products Recently
Approved and In Development Product Description Status DR-102 (Teva) Oral contraceptive
PhaseIII Nomac/E2 (Merck) Oral contraceptive PhaseIII Yaz Flex (Bayer) Oral
contraceptive Phase III Vaginal Ring (Actavis & Pop Council) Vaginal Ring
Phase II Nestragel™ (Antares & Pop Council) Topical gel contraceptive
Phase II Estelle (Actavis) Oral contraceptive Phase II Sources:
clinicaltrials.gov, accessed Feb 2014, BioPharm Insight by Infinata, July
2013, Company press releases Contraceptive Patch Products Product Description
Status Xulane™ (Mylan) Generic equivalent to Ortho Evra Same label as Evra
Launch announced Apr 2014 WAC price = $95.12/cycle Bay86-5016 (Bayer)
Transdermal Patch containing: Gestodene + EE 20ug/d Transparent patch US
Phase III completed Sep 2010 US NDA not submitted Approved in Europe, Feb2014
Other CHC Products in Development
|
|

|
24
Rule.pngProperty of Agile Therapeutics –06.26.2014 $41.53 $45.57 $50.35
$59.49 $68.57 $75.87 $81.88 $89.35 $94.44 $0.00 $10.00 $20.00 $30.00 $40.00
$50.00 $60.00 $70.00 $80.00 $90.00 $100.00 $110.00 2006 2007 2008 2009 2010
2011 2012 2013 2014 Avg. Price/Cycle for Branded Contraceptives ($WAC) Branded
Contraceptives Continue to Take Aggressive Price Increases Source: Price Rx
Select, as of Feb2014. *only includes price increases which occurred through
Feb 2014 Calculations include 13 leading branded contraceptive products. Avg.
Annual Price Increase 2006 2007 2008 2009 2010 2011 2012 2013 2014* 12.2%
9.7% 10.3% 17.3% 13.5% 11.6% 7.7% 8.5% 5.6%
|
|

|
25
Rule.pngProperty of Agile Therapeutics – 06.26.2014 Product Share
Allocations: Pre-and Post-Twirla Product Profile % of next 100 new patients
3% 1% 3% 14% 14% 15% 82% 67% 74% 17% 9% 0% 10% 20% 30% 40% 50% 60% 70% 80%
90% 100% Mean %Share of next 100 new patients Twirla Oral (pill) Vaginal ring
(NuvaRing) Contraceptive patch (Ortho-Evra) Pre-product profile share
allocations Share allocations after seeing AG200-15 AG200-15 Market Share
Calibrated for Overstatement Kantar Health Demand Calibration Model: Adjusts
for physician overstatement Source: Contraceptive Patch Assessment Study
(n=152 ObGyns), Kantar Health, Dec 2010 ObGyns Estimate Use of Twirla in 9%
of New Contraceptive Patients
|
|

|
26
Rule.pngProperty of Agile Therapeutics – 06.26.2014 Twirla has Significant
Peak Revenue Potential AG200-15 Annual Peak Sales Projection Based on Market
Share Potential 9% TRx Share (based on market research) $972 Million Revenue
Potential for each Market Share Point is Significant Sources: IMS NPA Dec
2013 and Wolters Kluwer Price Rx Select, Sep 2013 88 Million TRx (Total
Market, 2013) $88 per cycle (Avg. WAC, 2013) 1.4 cycles/TRx (Avg. Rx size,
2013) 1% $108 Million per 1% TRx share
|
|

|
27 Property of
Agile Therapeutics –06.26.2014 ObGyn Focus on Contraceptives Allows for Small
Sales Force of ~70 to 100 Representatives ObGyns and NP/PAs Account for ~70%
of U.S. Contraceptive Prescriptions Source: IMS NPA, TRx Volume by Prescriber
Type, 2013 Source: IMS NPA, TRX Volume by Category, 2010 Contraceptive TRx
Volume by Prescriber Type ObGyn TRx Volume by Category ObGyn/NP’s 69%
|
|

|
28
Rule.pngProperty of Agile Therapeutics –06.26.2014 Agile is Prepared for
Commercial-Scale Manufacturing Agile has an exclusive agreement with Corium
International, Inc. •Corium responsible for all aspects of manufacturing
•Substantial build-out of manufacturing facilities completed •Robust process
developed for commercial-scale manufacturing •>$10 Million investment by
Agile in commercial-scale equipment •Same process for clinical trials and
commercial materials
|
|

|
29
Rule.pngProperty of Agile Therapeutics –06.26.2014 Financial Profile
Background •Founded in 1997 •Approximately $121.2 Millionof funding from
inception to December 31, 2013 •Non-dilutive sources of capital –$15
Millionventure debt (Dec 2012) –$3.6 Millionfrom sale of state NOLs (Feb
2014) •$3.0 Millioncash on hand at 3/31/14 –$3.0 Million bridge financing
(Apr 2014) Use of proceeds •$55.0 Million gross proceeds (~$49.1 Millionnet
proceeds) –$31 Millionfor additional Phase 3 clinical trial for Twirla
–Completion of equipment validation and expansion of manufacturing
capabilities –Development of product candidate pipeline including line
extensions –Principal (beginning Feb 2015) and interest payments on term loan
continuing through Jul 2017
|
|

|
30 Property of
Agile Therapeutics –06.26.2014 Agile Corporate Accomplishments Jan
2014Elizabeth Garner, MD joined as Chief Medical Officer (CMO) Feb
2014Received $3.6 Million through New Jersey Technology Business Tax
Certificate Transfer (NOL) program Feb 2014Dan Shames, MD joined Scientific
Advisory Board (SAB) Former FDA Director, Division of Reproductive and
Urologic Products/CDER Mar 2014William McKee appointed to Board of Directors
Former CFO, Barr Pharmaceuticals, LLC Mar 2014Filed registration statement
(S-1) for proposed initial public offering (IPO) Mar 2014Katie MacFarlane,
PharmD joined as Chief Commercial Officer (CCO) May 2014Completed initial
public offering (IPO) for $55 Million Jun 2014Sixth U.S. patent granted on
Skinfusion®transdermal technology –to list in Orange Book
|
|

|
31 Property of
Agile Therapeutics –06.26.2014 Development Milestones Twirla Milestones 2014
2015 2016 Expected First Patient, First Visit (FPFV) FDA meeting, clear path
forward identified Anticipated validation of additional line (full commercial
capacity) Expected Last Patient, Last Visit (LPLV) FDA comments on protocol
received Line Extensions and New Products AG200-ER AG890 AG200-SP Program
design initiation, potential to progress into phase 3 in 2015 Patch
development required, potential to initiate phase 1 in 2015 Data analysis and
patch development required, clinical decision point in 2015 2014 2015 2016
|
|

|
32
Rule.pngProperty of Agile Therapeutics –06.26.2014 Agile Investment Thesis
•$4.2 Billion combined hormonal contraceptive (CHC) market •Significant Unmet
Need: No low-dose CHC patch on the market today Large Market Opportunity
•Over 1,500 women have received Twirla in clinical trials that showed
favorable safety and tolerability •Two completed randomized phase 3 trials
showed pregnancy rate comparable to comparator pills Significant Clinical
Experience •CRL and FDA communications provide clear guidance on path forward
with one single-arm trial •Potential for near-term approval in late 2016
Clear Regulatory Path •Wholly owned assets means company is free to partner
or sell •Company can market directly through focused sales force Multiple
Strategic Options •Deep experience in women’s health and contraceptive
products World Class Team •6 issued patents, protection to 2028
•Technological and manufacturing know-how High Barriers to Generic Entry
|
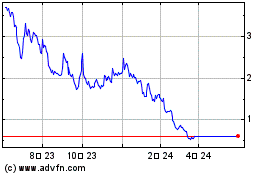
Agile Therapeutics (NASDAQ:AGRX)
과거 데이터 주식 차트
부터 6월(6) 2024 으로 7월(7) 2024
Agile Therapeutics (NASDAQ:AGRX)
과거 데이터 주식 차트
부터 7월(7) 2023 으로 7월(7) 2024